Setup of an osmosis plant and water softening plant
Water treatment usually consists of:
- Softening plant
- Reverse osmosis plant
- (increasingly: EDI, electrodeionization)
- (occasionally mixed bed)
- Permeate tank
- Pressure boosting

Risks in the water – What can happen?
Salts are dissolved in all natural water. The type and concentration of water constituents in drinking water varies depending on the origin of the water and the method of extraction.
The water ingredients can lead to the following problems:
Hardness components (calcium and magnesium salts).
Scale formation, lime scale due to calcium and magnesium hydrogen carbonate, corrosion potential
Heavy and non-ferrous metals, e.g. iron, manganese, copper Brownish-red coating formation, subsequent rusting
Silicates, silicic acid
White-gray, colored appearing, thin deposits
Chloride
Pitting corrosion
Evaporation residues Stains and coatings
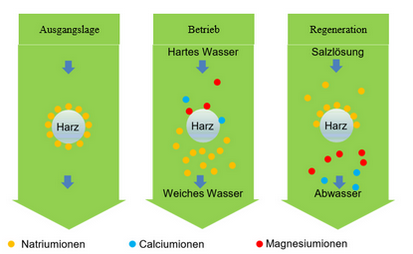
How water softeners work
The ion exchange process is based on a natural principle that can be found, for example, in porous zeolite rock. This uses the exchange of ions by releasing minerals into the water, while other minerals are removed from the water.
In softening plants, resins take over this function. The resin used is loaded with sodium ions in the initial position. To prevent stagnant germination , 3% Bio Resin is added to the resin.
If hard water meets the resin, the hardness formers will be Calcium and Magnesium against Sodium ions exchanged until the resin is saturated. The result is thus „Soft water“ erzeugt.
During regeneration, the resin is enriched again with sodium ions via salt tablets (salt = NaCl).
The special resin thus reloads itself with the sodium ions and in the course of this releases the stored calcium and magnesium ions into the wastewater.
Now new calcium and magnesium ions can come and in turn be exchanged for sodium ions. This creates a cycle.
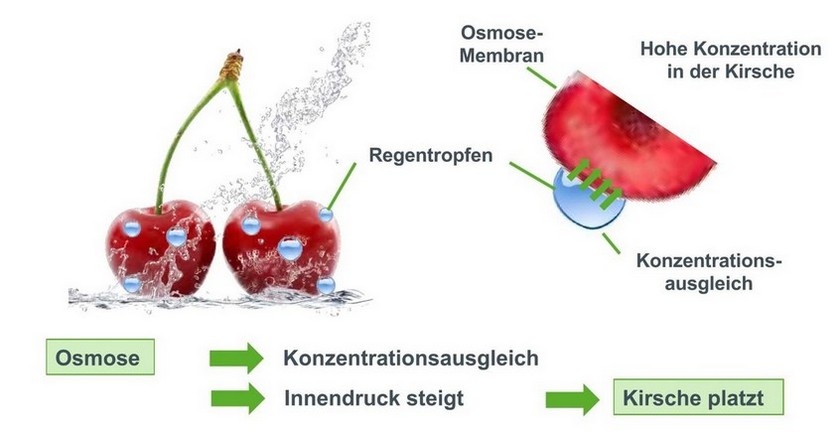
Reverse osmosis: How osmosis plants work
Osmosis – What is it?
Osmosis refers to the process of equalizing the concentration of two liquids through a semipermeable membrane. This process always occurs when two aqueous solutions with different ion concentrations are separated by a semi-permeable wall.
Physically, the ion solutions – which are separated from each other by membranes – always strive to achieve a concentration balance.
This means that ions want to move from the high concentration side to the lower concentration side.
Since the membrane is a barrier that the ions cannot easily pass through due to their molecular size, the smaller water molecules flow from the lower concentrated side to the higher concentrated side instead. (osmotic pressure)
Reverse osmosis process – Reverse osmosis plant
The reverse osmosis process was developed as early as the 1960s on behalf of NASA, which needed a drinking water recycling system for manned space flights.
In reverse osmosis technology, the osmosis principle is reversed. On the side with the high ion concentrations (tap water, raw water), a pressure is applied that forces the water in the other direction, namely to the pure water side with the lower concentration. The applied pressure is greater than the prevailing osmotic pressure.
The undesirable dissolved substances (e.g. hardness components, nitrate, silicic acid, residues of pesticides and medications, …) cannot pass through the ultrafine membrane due to their molecular size – on the pure water side, this means that almost exclusively water.
Since tap water with the substances it contains constantly flows in during operation, the substances retained by the membrane must be continuously removed to prevent the membrane from clogging. As a result, a reverse osmosis system produces wastewater (concentrate) in addition to the pure water, which contains the undesirable substances in increased concentration and which are flushed away.
„almost exclusively water: depending on the membrane, the retention rate is 95 – 99%.
This means that up to 5% pass through the membranes, even if they are in optimal condition.“
Water softening plant and reverse osmosis
A softening system that is monitored with sensors or is only regenerated according to quantity represents the first weak point! A breakthrough of calcium & magnesium can block the membranes of the reverse osmosis and thus leads to a decrease in performance.
Recommendation:
Use of Testomat 808 (Package 1)
A single-stage reverse osmosis provides conductivity values between 5 – 15 µS/cm. (Recommendation: Retrofit a second stage or EDI. Behind the second stage or EDI, the conductivity will be < 0.1 µS/cm.
Problem: Only when functioning properly, the impurities such as SiO2 are successfully retained. The retention rates do not reach 100%.
Recommendation:
In both cases, the retrofitting of a blending bed is an option. The blending bed can be monitored by a single meter or the silicate measuring panel and serves as a “police filter“.
„If Testomat 808 SiO2 is operated with an analysis interval of 4 hours, this results in 2,190 measurements per year. This is currently contrasted by the validation that takes place 2 x per year“
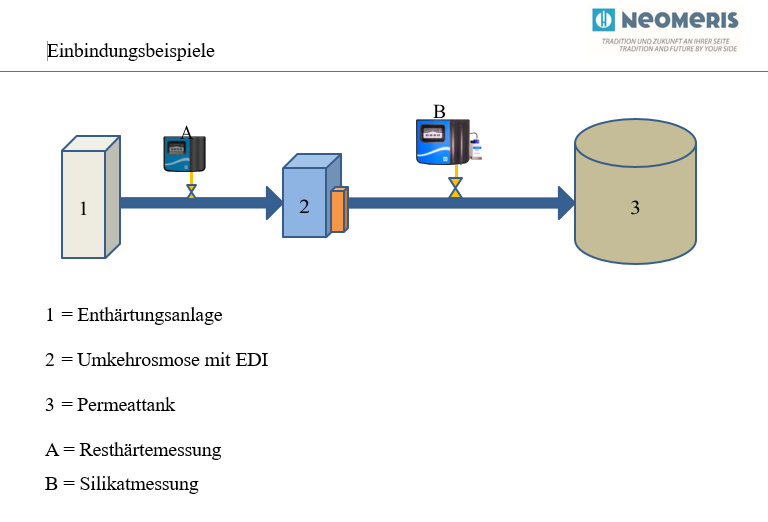


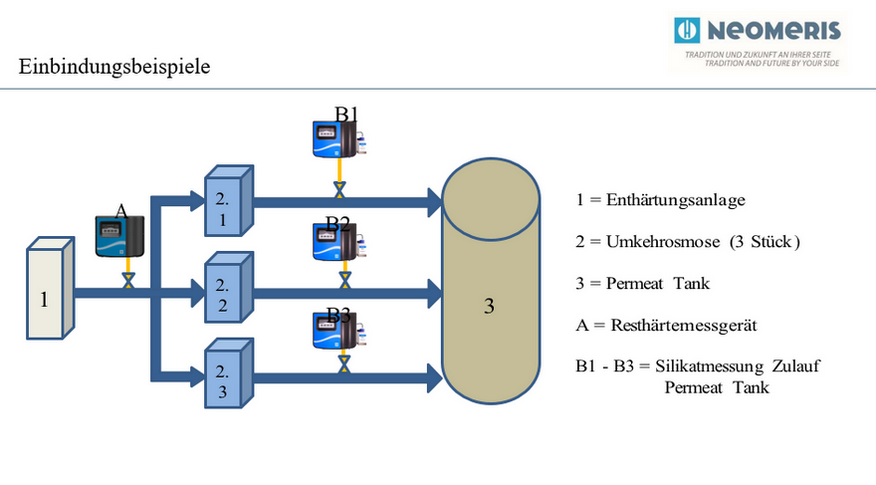
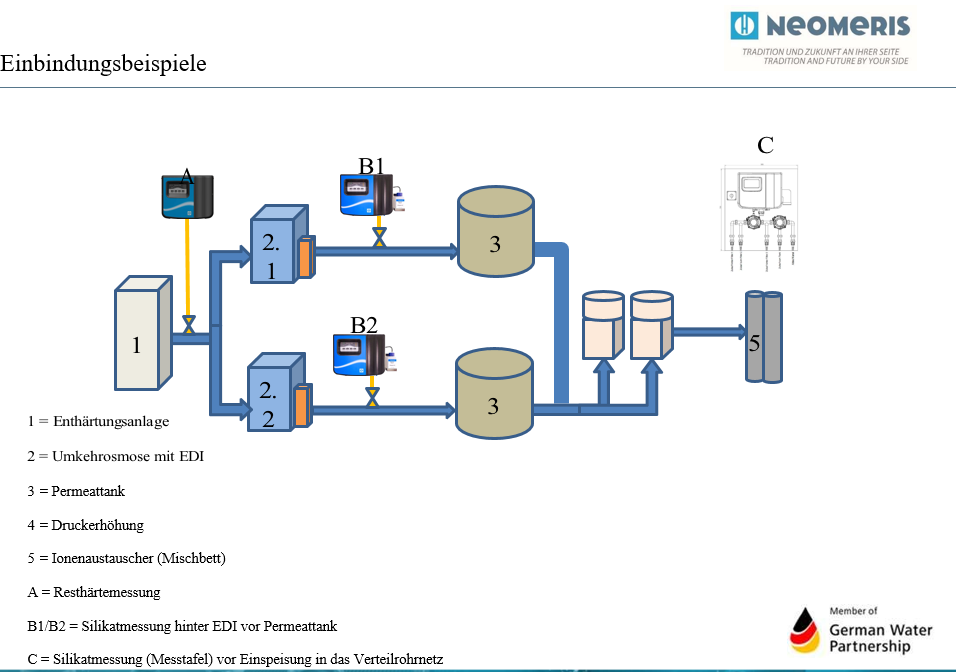

Notes osmosis plant – water softening plant
For the integration applies in principle:
- The pressure ratios of the installed systems must be taken into account in the design.
- The consumption behavior of the connected consumers must be taken into account, since the replenishment into the permeate tank is based on this (level monitoring).
- The measuring devices can be integrated in such a way that measurement only takes place if water is drawn off or replenished.
- Integration of the measuring devices by means of a 4-20 mA signal is possible and recommended.
Your advantage:
With a professionally installed water softener and reverse osmosis system, you can guarantee constant energy efficiency over the long term and optimally prevent system failures.
We will be pleased to help you with the planning of your water softening and reverse osmosis plant
as well as in the selection of a suitable company for the plant construction.
Please contact us.
Dipl.-Ing. (FH)
Karsten Hesse